- Record: found
- Abstract: found
- Article: found
A prospective, non-randomized, no placebo-controlled, phase Ib clinical trial to study the safety of the adipose derived stromal cells-stromal vascular fraction in idiopathic pulmonary fibrosis
Read this article at
Abstract
Introduction
Regenerative medicine and particular adult stem cells represent an alternative option with several fruitful therapeutic applications in patients suffering from chronic lung diseases including idiopathic pulmonary fibrosis (IPF). Nevertheless, lack of knowledge regarding the origin and the potential of mesenchymal stem cells (MSCs) to differentiate into fibroblasts has limited their use for the treatment of this dismal disease.
Patients and methods
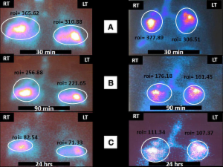
To this end, we conducted a phase Ib, non-randomized, clinical trial to study the safety of three endobronchial infusions of autologous adipose derived stromal cells (ADSCs)-stromal vascular fraction (SVF) (0.5 million cells per kgr of body weight per infusion) in patients with IPF (n=14) of mild to moderate disease severity (forced vital capacity –FVC>50% predicted value and diffusion lung capacity for carbon monoxide-DL CO>35% of predicted value). Our primary end-point was incidence of treatment emergent adverse events within 12 months. Alterations of functional, exercise capacity and quality of life parameters at serial time points (baseline, 6 and 12 months after first infusion) were exploratory secondary end-points.
Results
No cases of serious or clinically meaningful adverse events including short-term infusional toxicities as well as long-term ectopic tissue formation were recorded in all patients. Detailed safety monitoring through several time-points indicated that cell-treated patients did not deteriorate in both functional parameters and indicators of quality of life.
Related collections
Most cited references45
- Record: found
- Abstract: found
- Article: not found
A randomized, double-blind, placebo-controlled, dose-escalation study of intravenous adult human mesenchymal stem cells (prochymal) after acute myocardial infarction.
- Record: found
- Abstract: found
- Article: not found