- Record: found
- Abstract: found
- Article: found
Patterns of antimicrobial agent prescription in a sentinel population of canine and feline veterinary practices in the United Kingdom
Read this article at
Highlights
-
•
Antimicrobial agent prescription was monitored in a large UK population of cats and dogs over a 2 year period (2014–2016).
-
•
Systemic antimicrobial agents were prescribed more frequently to cats; topical prescription was more frequent in dogs.
-
•
A temporal reduction (2014–2016) in antimicrobial agent prescription was observed in both cats and dogs in this population.
-
•
Premises which prescribed antimicrobial agents commonly to cats generally also prescribed commonly to dogs.
-
•
The most frequently prescribed antibiotics were cefovecin in cats and clavulanic acid potentiated amoxicillin in dogs.
Abstract
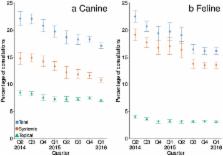
Antimicrobial resistance is an increasingly important global health threat and the use of antimicrobial agents is a key risk factor in its development. This study describes antimicrobial agent prescription (AAP) patterns over a 2 year period using electronic health records (EHRs) from booked consultations in a network of 457 sentinel veterinary premises in the United Kingdom. A semi-automated classification methodology was used to map practitioner defined product codes in 918,333 EHRs from 413,870 dogs and 352,730 EHRs from 200,541 cats, including 289,789 AAPs. AAP as a proportion of total booked consultations was more frequent in dogs (18.8%, 95% confidence interval, CI, 18.2–19.4) than cats (17.5%, 95% CI 16.9–18.1). Prescription of topical antimicrobial agents was more frequent in dogs (7.4%, 95% CI 7.2–7.7) than cats (3.2%, 95% CI 3.1–3.3), whilst prescription of systemic antimicrobial agents was more frequent in cats (14.8%, 95% CI 14.2–15.4) than dogs (12.2%, 95% CI 11.7–12.7). A decreasing temporal pattern was identified for prescription of systemic antimicrobial agents in dogs and cats. Premises which prescribed antimicrobial agents frequently for dogs also prescribed frequently for cats. AAP was most frequent during pruritus consultations in dogs and trauma consultations in cats. Clavulanic acid potentiated amoxicillin was the most frequently prescribed antimicrobial agent in dogs (28.6% of prescriptions, 95% CI 27.4–29.8), whereas cefovecin, a third generation cephalosporin, was the most frequently prescribed antimicrobial agent in cats (36.2%, 95% CI 33.9–38.5). This study demonstrated patterns in AAP over time and for different conditions in a population of companion animals in the United Kingdom.
Related collections
Most cited references18
- Record: found
- Abstract: found
- Article: not found
Possible Transmission of mcr-1–Harboring Escherichia coli between Companion Animals and Human
- Record: found
- Abstract: found
- Article: not found
Characterisation of antimicrobial usage in cats and dogs attending UK primary care companion animal veterinary practices.
- Record: found
- Abstract: found
- Article: not found