- Record: found
- Abstract: found
- Article: found
The Global Burden of Disease Study 2010: Interpretation and Implications for the Neglected Tropical Diseases
research-article
Peter J. Hotez
1
,
2
,
3
,
* ,
Miriam Alvarado
4 ,
María-Gloria Basáñez
5 ,
Ian Bolliger
4 ,
Rupert Bourne
6 ,
Michel Boussinesq
7 ,
Simon J. Brooker
8 ,
Ami Shah Brown
9 ,
Geoffrey Buckle
10 ,
Christine M. Budke
11 ,
Hélène Carabin
12 ,
Luc E. Coffeng
4
,
13 ,
Eric M. Fèvre
14
,
15 ,
Thomas Fürst
5
,
16
,
17 ,
Yara A. Halasa
18 ,
Rashmi Jasrasaria
4 ,
Nicole E. Johns
4 ,
Jennifer Keiser
16
,
17 ,
Charles H. King
19 ,
Rafael Lozano
4 ,
Michele E. Murdoch
20 ,
Simon O'Hanlon
5 ,
Sébastien D. S. Pion
7 ,
Rachel L. Pullan
8 ,
Kapa D. Ramaiah
21 ,
Thomas Roberts
22 ,
Donald S. Shepard
18 ,
Jennifer L. Smith
8 ,
Wilma A. Stolk
13 ,
Eduardo A. Undurraga
18 ,
Jürg Utzinger
16
,
17 ,
Mengru Wang
4 ,
Christopher J. L. Murray
4
,
* ,
Mohsen Naghavi
4
,
*
24 July 2014
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Introduction
The publication of the Global Burden of Disease Study 2010 (GBD 2010) and the accompanying
collection of Lancet articles in December 2012 provided the most comprehensive attempt
to quantify the burden of almost 300 diseases, injuries, and risk factors, including
neglected tropical diseases (NTDs) [1]–[3]. The disability-adjusted life year (DALY),
the metric used in the GBD 2010, is a tool which may be used to assess and compare
the relative impact of a number of diseases locally and globally [4]–[6]. Table 1
lists the major NTDs as defined by the World Health Organization (WHO) [7] and their
estimated DALYs [1]. With a few exceptions, most of the NTDs currently listed by the
WHO [7] or those on the expanded list from PLOS Neglected Tropical Diseases
[8] are disablers rather than killers, so the DALY estimates represent one of the
few metrics available that could fully embrace the chronic effects of these infections.
10.1371/journal.pntd.0002865.t001
Table 1
Estimated DALYs (in millions) of the NTDs from the Global Burden of Disease Study
2010.
Disease
DALYs from GBD 2010 (numbers in parentheses indicate 95% confidence intervals) [1]
NTDs
26.06 (20.30–35.12)
Intestinal nematode infections
5.19 (2.98–8.81)
Hookworm disease
3.23 (1.70–5.73)
Ascariasis
1.32 (0.71–2.35)
Trichuriasis
0.64 (0.35–1.06)
Leishmaniasis
3.32 (2.18–4.90)
Schistosomiasis
3.31 (1.70–6.26)
Lymphatic filariasis
2.78 (1.8–4.00)
Food-borne trematodiases
1.88 (0.70–4.84)
Rabies
1.46 ((0.85–2.66)
Dengue
0.83 (0.34–1.41)
African trypanosomiasis
0.56 (0.08–1.77)
Chagas disease
0.55 (0.27–1.05)
Cysticercosis
0.50 (0.38–0.66)
Onchocerciasis
0.49 (0.36–0.66)
Trachoma
0.33 (0.24–0.44)
Echinococcosis
0.14 (0.07–0.29)
Yellow fever
<0.001
Other NTDs
*
4.72 (3.53–6.35)
* Relapsing fevers, typhus fever, spotted fever, Q fever, other rickettsioses, other
mosquito-borne viral fevers, unspecified arthropod-borne viral fever, arenaviral haemorrhagic
fever, toxoplasmosis, unspecified protozoal disease, taeniasis, diphyllobothriasis
and sparganosis, other cestode infections, dracunculiasis, trichinellosis, strongyloidiasis,
enterobiasis, and other helminthiases.
Even DALYs, however, do not tell the complete story of the harmful effects from NTDs.
Some of the specific and potential shortcomings of GBD 2010 have been highlighted
elsewhere [9]. Furthermore, DALYs measure only direct health loss and, for example,
do not consider the economic impact of the NTDs that results from detrimental effects
on school attendance and child development, agriculture (especially from zoonotic
NTDs), and overall economic productivity [10], [11]. Nor do DALYs account for direct
costs of treatment, surveillance, and prevention measures. Yet, economic impact has
emerged as an essential feature of the NTDs, which may trap people in a cycle of poverty
and disease [10]–[12]. Additional aspects not considered by the DALY metrics are the
important elements of social stigma for many of the NTDs and the spillover effects
to family and community members [13], [14], loss of tourism [15], and health system
overload (e.g., during dengue outbreaks). Ultimately NTD control and elimination efforts
could produce social and economic benefits not necessarily reflected in the DALY metrics,
especially among the most affected poor communities [11].
Variations in DALYs
Despite the importance of the concept of disease burden and disability to the NTD
community, assigning DALYs or related metrics to each NTD has been a bit of a roller-coaster
ride over the past decade and may continue to be for many years to come. Significant
variations in ascribing DALYs to the NTDs are due to many factors, including data
scarcity and inherent difficulties in accurately estimating the number of individuals
at risk, the number of incident cases, the number of prevalent cases, and, among these,
the duration of the infection. Challenges also include uncertainty about the relationship
between acute and chronic infections and their link to specific morbidities, duration
of morbidity, and the proportion of the population infected or with morbidities that
are treated versus untreated. An additional challenge is to obtain all of the aforementioned
values stratified by age and gender, data which are seldom available for NTDs. Moreover,
the affordable diagnostic tools typically used to measure NTDs in resource-constrained
settings are inaccurate and many sequelae (i.e., morbidities) of NTDs are nonspecific,
making it difficult to attribute them to a particular infection or risk factor. For
several NTDs, controversies remain regarding what proportion of a sequelae should
be ascribed to different infections or diseases. An extreme example is the case of
schistosomiasis, for which disease burden estimates over the past decade have ranged
from 1.7 million DALYs to as many as 56 million DALYs, depending on whether higher
disease prevalence estimates are considered and if specific chronic morbidities are
attributed to this NTD [12]. The variation is also due to continuous refinement of
definitions and methodologies for burden estimation, which affects the estimates for
all diseases, injuries, and risk factors and further complicates the comparison of
different GBD versions. Among the furthest-reaching methodological alterations of
GBD 2010 are the shift from incidence- to prevalence-based DALYs, the abandonment
of age weighting and discounting, the application of refined reference life tables
and disability weights, and the introduction of comorbidity adjustments [16].
Some of the greatest variation in the disease burden estimates over the past decade
has been observed among the three major intestinal nematode infections (also known
as soil-transmitted helminthiases, i.e., ascariasis, hookworm disease, and trichuriasis)
as well as in schistosomiasis. A key reason for this wide variation is the fact that
these helminth infections are among the most common infections of humankind [17]–[19],
so small variations in an assigned disability weight become amplified by the hundreds
of millions of people estimated to harbor these parasites. Another reason for variations
in some burden estimates is due to how GBD 2010 uniquely classified certain diseases
or groups of diseases. A prominent example was the decision to combine the burdens
of cystic echinococcosis and alveolar echinococcosis into a single estimate (i.e.,
echinococcosis). This was a questionable decision seeing that the two parasites have
different life cycles, geographic distributions, and clinical outcomes. Future iterations
of the GBD will therefore need to consider reporting these estimates as separate conditions,
paying greater attention to the unique attributes of the individual parasites.
Overall, the NTD community was dismayed by the previous WHO estimates between 1999
and 2004 [20], which assigned DALYs that were equivalent to conditions of comparatively
minor global health importance for major diseases such as schistosomiasis [21]. At
the other extreme, the higher DALY estimates for NTDs elevate the status of these
diseases to a level at which they could be thought of as the fourth leg to a table
built on HIV/AIDS, tuberculosis, and malaria [22]. The GBD 2010 is an ambitious attempt
to resolve some of the differences between earlier estimates (including use of strictly
comparable data and methods for 1990, 2005, and 2010) and to provide a first attempt
at estimating the disease burden of cysticercosis, echinococcosis, and rabies as part
of the largest ever burden of disease study [1]–[3]. The GBD 2010 also provides first-time
disease burden estimates for amebiasis, cryptosporidiosis, trichomoniasis, scabies,
fungal skin infections, and venomous animal contact (including snake bite), although
they are not listed under the NTD category (Table 2) [1]–[3]. One surprising finding
from these estimates was the huge disease burden that results from cryptosporidiosis
among young children. Together, the NTDs listed in Table 1 and those in Table 2 add
up to almost 48 million DALYs. This number is comparable to tuberculosis (49 million)
and is more than half of the global burden of two of the world's major diseases, malaria
(83 million) and HIV/AIDS (82 million). However, these comparisons must be conducted
with great care given the large variation in the quantity and quality of epidemiological
data currently available across the world.
10.1371/journal.pntd.0002865.t002
Table 2
Other NTDs in the Global Burden of Disease Study 2010 not listed in the “NTD and malaria”
category.1
Disease
DALYs from GBD 2010 in millions (numbers in parentheses indicate 95% confidence intervals)
[1]
Cryptosporidiosis
8.37 (6.52–10.35)
Cholera
4.46 (3.34–5.80)
Animal contact (venomous)
2.72 (1.54–4.80)
Amebiasis
2.24 (1.73–2.84)
Fungal skin diseases
2.30 (0.72–5.27)
Scabies
1.58 (0.80–2.79)
Trichomoniasis
0.17 (0.01–0.53)
Leprosy
0.006 (0.002–0.11)
Total
21.84
Total of NTDs in
Table 1
(from GBD 2010) and NTDs in
Table 2
47.90
1
The table provides numbers of DALYs in millions as calculated in GBD 2010 [1]. The
diseases are not listed as NTDs in GBD 2010 and, with the exception of leprosy, these
diseases are also not on the WHO list of 17 NTDs [5]. However, these conditions (as
well as some other diarrheal diseases) are considered by PLOS Neglected Tropical Diseases
[6].
Killers and Disablers
Some of the details of the new disease burden estimates for NTDs are summarized in
Table 3, while the total number of estimated cases is summarized in Table 4. Briefly,
as stated by Murray et al. (2012), “DALYs are the sum of two components: years of
life lost due to premature mortality (YLLs) and years lived with disability (YLDs)”
[1]. For many of the major NTDs, including hookworm disease and the other intestinal
nematode infections, schistosomiasis, food-borne trematodiases, onchocerciasis, cysticercosis,
and trachoma, most (and in some cases all) of the reported DALYs result from YLDs
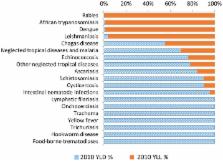
(i.e., disability, not deaths) (Figure 1). These NTDs are genuinely not thought of
as killer diseases, although it has been noted that some disabling NTDs such as onchocerciasis,
cysticercosis, and food-borne trematodiases cause excess mortality associated with
blindness, heavy infection in sighted individuals, hydrocephalus, stroke, gliomas,
ectopic infections, cholangiocarcinoma, and other (yet unmeasured) factors [23]–[26].
An added feature about the publication of the YLDs from the NTDs was the listing of
the specific sequelae that were considered in deriving these estimates [3], which
allows comparability across studies.
10.1371/journal.pntd.0002865.g001
Figure 1
Fractions of YLD and YLL (as components of DALYs) for each of the NTDs. Also included
in this graph are “other NTDs.”
10.1371/journal.pntd.0002865.t003
Table 3
DALYs, YLDs, YLLs, and deaths from NTDs from the Global Burden of Disease Study.
Disease
DALYs in millions [1]
DALY rank
YLDs in millions [3]
YLD rank
YLLs in millions [1]–[3]
YLL rank
Deaths [2]
Death rank
All NTDs
26.06
18.22
7.90
152,000
Intestinal nematode infections
5.19
1
4.98
1
0.20
7
2,700
7
Hookworm disease
3.23
4
3.23
2
0
-
-
Ascariasis
1.32
8
1.11
6
0.20
7
2,700
7
Trichuriasis
0.64
10
0.64
7
0
-
-
Leishmaniasis
3.32
2
0.12
12
3.19
1
51,600
1
Schistosomiasis
3.31
3
2.99
3
0.32
5
11,700
4
Lymphatic filariasis
2.78
5
2.77
4
0
-
-
Food-borne trematodiases
1.88
6
1.87
5
0
-
-
Rabies
1.46
7
<0.01
16
1.46
2
26,400
2
Dengue
0.83
9
0.01
15
0.81
3
14,700
3
African trypanosomiasis
0.56
11
0.08
14
0.55
4
9,100
6
Chagas disease
0.55
12
0.30
11
0.24
6
10,300
5
Cysticercosis
0.50
13
0.46
9
0.05
8
1,200
8
Onchocerciasis
0.49
14
0.49
8
0
-
-
Trachoma
0.33
15
0.33
10
0
-
-
Echinococcosis
0.14
16
0.11
13
0.03
9
1,200
8
Yellow fever
<0.001
17
<0.01
16
<0.01
10
-
Other NTDs
4.72
-
3.69
-
1.03
-
23,700
10.1371/journal.pntd.0002865.t004
Table 4
Expected number of cases in 2010 and 95% confidence intervals of the neglected tropical
diseases (mean and uncertainty) as extrapolated from the Global Burden of Disease
Study 2010.
Disease
Number of cases
95% confidence intervals
Selected comments
Ascariasis1
819 million
772–892 million
Total number of cases
Trichuriasis1
465 million
430–508 million
Total number of cases
Hookworm disease1
439 million
406–480 million
Total number of cases
Schistosomiasis
252 million
252–252 million
Total number of cases
Onchocerciasis
30.4 million
27.3–33.6 million
Total number of cases with adult worms*
Lymphatic filariasis
36 million
34–39 million
Lymphedema and/or hydrocele only
Food-borne trematodiases
16 million
7–41 million
Heavy and cerebral infections only
Cutaneous leishmaniasis
10 million
8–13 million
Total number of cases
Chagas disease
7.5 million
2.5–12.4 million
Symptomatic cases only
Trachoma
4.4 million
3.5–5.5 million
Low vision and blindness cases only
Cysticercosis
1.4 million
1.3–1.6 million
Epilepsy cases only
Echinococcosis
1.1 million
0.6–2.1 million
Symptomatic liver, lung, and central nervous system cases only
Dengue
179,000 cases
109,000–299,000
Incident (acute) symptomatic cases only
Visceral leishmaniasis
76,000 cases
61,000–93,500
Total number of cases
African trypanosomiasis
37,000 cases
9,000–106,000
Symptomatic cases only
Rabies
1,100 cases
600–2,000
Incident cases
Yellow fever
100 cases
0–100 cases
Incident cases
* This number includes 14.6 million people (13.2–16.1 million) with detectable skin
microfilariae.
1
These are updated estimates recently published in Pullan et al. [27].
According to the GBD 2010 estimations, intestinal nematode infections rank first in
the list of the NTDs for which a DALY was estimated [27]. Among intestinal nematodes,
hookworm disease was estimated as having the largest YLDs (and 62% of the DALYs).
This large contribution of hookworm disease to the YLDs of nematodes comes from the
inclusion of recent information linking hookworm disease to moderate and severe anemia
across several different populations, including children and pregnant women [28],
[29]. On the other hand, important comorbidity effects resulting from hookworm disease
and malaria coinfections [30]–[32] and the deaths from these conditions were attributed
to malaria in the GBD 2010, reducing the apparent YLLs of hookworm infections.
Schistosomiasis was estimated to rank second in terms of YLDs (and right behind the
intestinal nematode infections in terms of prevalence). Schistosomiasis was one of
the NTDs that generated the most controversy and debate in the GBD 2010. Since 2005,
important information has been generated about the effects of schistosomiasis that
result in chronic pain, inflammation, malnutrition, and exercise intolerance, among
other morbid sequelae [12], [21], [33], which under some scenarios generated DALY
estimates that exceeded those of malaria or other better-known conditions [12]. However,
many of these aspects were not accepted into the GBD 2010, in part because of disagreements
about the long-term health importance and actual YLLs caused by these elements. Fueling
the schistosomiasis controversy even further were previously published annual mortality
estimates for schistosomiasis (i.e., 280,000 in Africa alone) [33] suggesting that
the number of people killed from this disease was at least 20 times higher than indicated
in GBD 2010 [34]. In addition, there is new information on the links between female
urogenital schistosomiasis and the risk of acquiring HIV/AIDS [35]. The discussions
surrounding the burden of schistosomiasis may just be the start of future investigations
on how to best attribute parts of the burden of chronic diseases and sequelae to NTDs.
Only through such debates will the estimations of the burden of disease further improve.
There are two major NTDs linked to blindness—trachoma and onchocerciasis. For trachoma,
the DALYs only consider disease due to active infection and do not consider blindness
that exists even after removal of the infection. For onchocerciasis, the DALYs do
not consider the excess mortality due to blindness [23] and likely underestimate the
effects of onchocercal skin disease. Furthermore, the onchocerciasis estimates have
ignored the burden in the Americas and low-endemic African countries, which may now
be relatively small compared to the burden in Africa but was not negligible in 1990.
Hence, in both instances the disease burdens from blinding NTDs may represent underestimates.
Finally, in terms of YLDs, important “newcomers” on the GBD scene were the food-borne
trematodiases, cysticercosis, and echinococcosis, which must now be recognized as
important causes of global disability. Still, no deaths were ascribed to either clonorchiasis
or opisthorchiasis (two of the key food-borne trematode infections) in the GBD 2010,
despite the strong evidence base linking these liver fluke infections to cholangiocarcinoma
in Southeast Asia and elsewhere [36], [37]. Similarly, the YLLs from cysticercosis
are most likely underestimated. Indeed, a recent systematic review of the literature
showed the proportion of neurocysticercosis patients under care who died during their
follow-up could vary from 0.9% to 18.5% [27]. Mostly due to a lack of available data
on a global scale, the current estimate for cysticercosis is limited to its role in
epilepsy in endemic countries and does not yet include the role of this infection
in causing severe chronic headaches and hydrocephalus, depressive disorders, stroke,
gliomas, and other neurological sequelae [24].
Among the killer NTDs, almost all of the DALYs due to diseases such as rabies, dengue,
and African trypanosomiasis resulted from YLLs, and practically no disability was
associated with nonlethal effects from these conditions (YLDs) (Figure 1). However,
for dengue, considerable evidence now points to a potentially higher percentage of
DALYs due to YLDs (∼25%) as a result of underreporting of nonfatal cases [38], [39].
Similarly, for leishmaniasis the DALY estimates mostly considered the large number
of deaths resulting from visceral leishmaniasis but included virtually nothing from
the disability of cutaneous leishmaniasis. This finding is a debatable point given
the evidence linking disfiguring cutaneous (and mucocutaneous) leishmaniasis on the
face to stigma and its impact on girls and women [40]. In addition, for African trypanosomiasis
there is also a long-term disease burden resulting from nonfatal consequences, including
those suffered by survivors who are eventually treated [41]. Chagas disease was one
of the important NTDs whose DALYs were roughly equally distributed between YLDs and
YLLs.
Trends
Figure 2 depicts the ranking of the different NTDs in 1990 as compared to 2010. Although
the estimates for both years stem from GBD 2010 and are therefore extrapolated by
using the same methodology, they must be interpreted with great care given that the
accuracy of the underlying data may have changed through time, with more accurate
diagnostic tests becoming available in recent years. The survey locations for frequency
data may also have varied between the two periods.
10.1371/journal.pntd.0002865.g002
Figure 2
Global trends in DALYs from NTDs, 1990 to 2010.
*Estimation of percent (%) change is not from the means. Each metric in this figure
is estimated on 1000 times in the modeling process, and then causes that have a high
degree of uncertainty in their draw estimates can have skewed % change results. Abbreviations:
UI, unit interval.
As shown in Figure 2, ascariasis exhibited the largest decrease in DALYs, possibly
as a consequence of deworming and socioeconomic development, although it could also
reflect the fact that many follow-up studies may have been conducted in areas where
such control programs took place. In addition, ascariasis exhibited the greatest decrease
in rank, whereas the rankings for trichuriasis and hookworm disease remained constant.
The basis for this difference among the intestinal nematode infections is not known,
although it may be related to the differential susceptibility of the different helminth
species to benzimidazole anthelmintics [42]. It is anticipated that helminth control
through mass drug administration and improved access to clean water and sanitation
may alter epidemiologic patterns and disease prevalence in the coming years [43].
African trypanosomiasis and rabies (and some other NTDs) were also greatly diminished,
the former possibly due to increased access to public health control in association
with the resolution of some civil and international conflicts in sub-Saharan Africa
[44]. In contrast, DALY estimates for schistosomiasis, lymphatic filariasis, and trachoma
appear to have increased over the past 20 years. The underlying bases for these increases
include population growth, ecological transformations (e.g., construction of large
dams and irrigation systems), and possibly increased surveillance, although it is
anticipated that as integrated parasitic disease control and preventive chemotherapy
initiatives progress and access to clean water and sanitation increases, we should
witness a reduction in several of these disease burden estimates in future years [43].
For dengue, urbanization and increases in global commerce and travel contribute to
the emergence of this important disease [45], [46], but increased access to diagnostic
tools may also play a role. Since the publication of the GBD 2010, a new estimate
suggests that as many as 390 million cases of dengue infections now occur annually
[47], more than three times the previous estimates by the WHO.
Geographic Distribution
Comparison in the geographical distribution of NTDs must also be conducted with great
care since the quality and quantity of data available will depend on where epidemiological
studies have been conducted. In addition, within each country, the reported country-level
DALYs may be based on surveys conducted specifically in areas where an infection is
known to be endemic, which may increase their relative importance as compared to countries
where surveys have not been conducted due to a lack of funding or have been conducted
in both endemic and nonendemic areas of the country. It is also important to emphasize
that many NTDs are of local or of focal importance, often affecting marginalized populations
who may not be recognized as national priorities [48]. However, keeping these limitations
in mind, the GBD 2010 suggests that there exists an extensive geographic distribution
of the NTDs, with sub-Saharan Africa representing the highest DALY rate per 100,000
individuals from NTDs—in part because of their high prevalence together with coinfections
that result from hookworm disease, schistosomiasis, onchocerciasis, and African trypanosomiasis
[1]. Oceania also has a disproportionate share of NTDs (especially from hookworm disease
in Papua New Guinea), as does Southeast Asia, South Asia, and tropical Latin America
[1]. Overall the largest (net) number of DALYs from NTDs occurs in Asia (Figure 3).
It has been noted that the largest number of cases of many of the high-burden NTDs
actually occur in the large emerging-market Asian countries such as China, India,
and Indonesia, as well as other countries of the group of 20 (G20) nations [49].
10.1371/journal.pntd.0002865.g003
Figure 3
DALYs: Number by disease and for the 21 regions in 2010 (in thousands).
In many Latin American countries, Chagas disease is the predominant NTD. Exceptions
are several countries where either intestinal nematode infections predominate (e.g.,
Colombia, Ecuador, and Venezuela) or Chagas may be underreported, and Haiti and the
Dominican Republic, where dengue is the largest source of DALYs. In Bolivia and Peru,
food-borne trematodiases rank closely with Chagas disease as the leading NTDs, while
emerging information about Chagas disease in the United States [50] may eventually
make it an important NTD there as well. Schistosomiasis is the predominant NTD among
sub-Saharan African countries, except in selected nations where leishmaniasis (e.g.,
Sudan), African trypanosomiasis (e.g., Democratic Republic of the Congo, Central African
Republic, and Chad), onchocerciasis (e.g., Cameroon), lymphatic filariasis (e.g.,
Senegal and Guinea-Bissau), intestinal nematode infections (South Africa, Botswana,
and Namibia), or rabies (Niger) rank higher. In the Middle East, leishmaniasis is
an important NTD, while rabies is the predominant NTD in Afghanistan. In Asia, leishmaniasis
is the leading NTD in India; food-borne trematodiases predominate in China, North
Korea, and Japan; and intestinal nematode infections are the leading NTDs in much
of Southeast Asia (with the exception of dengue in Lao PDR) and Papua New Guinea.
Missing in Action
There remain some important NTDs for which there are no or limited published disease-burden
estimates. These include strongyloidiasis [51], toxocariasis [52], and loiasis, which
are among the most common parasitic nematode infections worldwide, as well as toxoplasmosis
[53], an important maternal-child protozoan infection that has recently been linked
to schizophrenia in immune-competent people and to issues of mental health; leptospirosis,
a major bacterial infection; and podoconiosis, a noninfectious condition. In order
to estimate the burden subsumed and named as “other NTDs”, the respective cases of
death were modeled by using a Cause of Death Ensemble model (CODEm) tool [2], [54],
and then the ratio of YLLs to YLDs as derived from the rest of the NTDs was applied
to extrapolate the respective YLDs.
Concluding Statements and Future Directions
An important overriding conclusion of the GBD 2010 is the apparent global shift away
from communicable to noncommunicable diseases (NCDs) [1], [55]. Such a conclusion
must be tempered by the knowledge that many NTDs are actually underlying causes of
the so-called NCDs. In 2008, several NCDs were described, including cancer, cardiovascular
disease, and liver disease, that result from chronic long-standing NTDs or from past
infections with NTDs such as cysticercosis [56]. With regards to cancer, a new review
has identified a substantial burden that can be attributed to infectious diseases
[57]. These estimates suggest that, globally, 16% of cancers are caused by underlying
infectious agents, and in some developing regions such as sub-Saharan Africa, almost
one-third of cancers are caused by infections [57]. In terms of the NTDs, it is known
that Schistosoma haematobium (the cause of urogenital schistosomiasis) and three of
the major liver flukes—Opisthorchis viverrini, O. felineus, and Clonorchis sinensis—are
potent carcinogens responsible for a substantial but largely unknown burden of bladder
cancer and cholangiocarcinoma, respectively [36], [58], [59]. The burden of cardiovascular
disease attributed to NTDs has been recently summarized [60], as have some interesting
links between NTDs and chronic liver disease [61] and between onchocerciasis and epilepsy
[62]. As new information is obtained, the number of NCD YLLs and YLDs attributed to
NTDs will almost certainly increase.
The GBD 2010 is not intended to be the final word on the global disease burden resulting
from NTDs. Additional research is needed for almost all of the NTDs, and it is expected
that as new information becomes available it can be incorporated into new DALY estimates.
For example, the annual number of officially reported dengue cases in eight endemic
countries in the Americas and Asia (574,000) is almost three times the episodes estimated
by GBD 2010 (Table 4) [63]. Other important examples include the nonlethal consequences
of African trypanosomiasis, dengue, and leishmaniasis that will add a larger YLD component
to disease burdens for these conditions, as well as the deaths that result from cysticercosis,
food-borne trematodiases, hookworm disease, onchocerciasis, and schistosomiasis, among
others, which will add YLLs. The GBD 2010 will be updated regularly, which might also
allow epidemiologists and policy makers to observe spatiotemporal and presumably declining
trends in ascariasis, African trypanosomiasis, lymphatic filariasis, onchocerciasis,
trachoma, and possibly other NTDs as a result of preventive chemotherapy and other
control interventions. In so doing, a sincere hope is that the GBD 2010 can become
a living and breathing document with the flexibility to adapt and change and can ultimately
resolve discrepancies and controversies on the true disease burden resulting from
NTDs and diseases, injuries, and risk factors.
Related collections
Most cited references42
- Record: found
- Abstract: found
- Article: not found
Schistosomiasis and water resources development: systematic review, meta-analysis, and estimates of people at risk.
Peter Steinmann, Jennifer Keiser, Robert Bos … (2006)
- Record: found
- Abstract: not found
- Article: not found
GBD 2010: design, definitions, and metrics.
Christopher Murray, Majid Ezzati, Abraham Flaxman … (2012)
- Record: found
- Abstract: found
- Article: not found
Reassessment of the cost of chronic helmintic infection: a meta-analysis of disability-related outcomes in endemic schistosomiasis.
Charles King, Katherine Dickman, Daniel J Tisch (2005)