- Record: found
- Abstract: found
- Article: found
Performance of point-of-care urine test in diagnosing tuberculosis suspects with and without HIV infection in selected peripheral health settings of Addis Ababa, Ethiopia
Read this article at
Abstract
Background
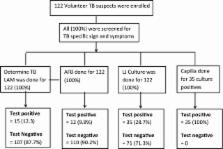
There are few rapid point-of-care tests (POCT) for tuberculosis (TB) for use in resource-constrained settings with high levels of human immunodeficiency virus (HIV). This hinders early tuberculosis (TB) treatment. This cross-sectional study evaluates the recently developed urine Determine tuberculosis lipoarabinomannan (TB LAM) antigen test. A total of 122 participants with signs and symptoms of TB, including 21 (17.1%) participants positive for HIV, were enrolled from September 2011 to March 2012 at three selected health centers in Addis Ababa, Ethiopia. Blood, sputum and urine samples were collected. Löwenstein-Jensen (LJ) solid culture was used as a gold standard to evaluate the performance of the Determine TB LAM antigen test. Data were analyzed using STATA (Statacorp LP, USA).
Results
Of the 122 participants with suspected TB, 35 (28.7%) had TB confirmed bacteriologically by LJ culture. The overall sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of Determine TB LAM (for both HIV-positive and HIV-negative participants) was 37.1% (95% CI 21.5–55.1), 97.7% (95% CI 91.9–99.7), 86.7% (95% CI 59.5–98.3) and 79.4% (95% CI 70.5–86.6), respectively. However, in participants who were co-infected with TB and HIV, sensitivity, specificity, PPV and NPV were 55.6% (95% CI 21.2–86.3), 100% (95% CI 73.5–100), 100% (95% CI 47.8–100) and 75.0% (95% CI 47.6–92.7). Moreover, the level of immunosuppression of the HIV-infected TB patients was found to have a significant association with the performance of Determine TB LAM (χ 2 = 7.89, p = 0.002).
Related collections
Most cited references18
- Record: found
- Abstract: found
- Article: not found
Meta-analysis: new tests for the diagnosis of latent tuberculosis infection: areas of uncertainty and recommendations for research.
- Record: found
- Abstract: found
- Article: not found
Diagnostic accuracy of a low-cost, urine antigen, point-of-care screening assay for HIV-associated pulmonary tuberculosis before antiretroviral therapy: a descriptive study

- Record: found
- Abstract: found
- Article: found