- Record: found
- Abstract: found
- Article: not found
A novel coronavirus outbreak of global health concern
discussion
24 January 2020
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
In December, 2019, Wuhan, Hubei province, China, became the centre of an outbreak
of pneumonia of unknown cause, which raised intense attention not only within China
but internationally. Chinese health authorities did an immediate investigation to
characterise and control the disease, including isolation of people suspected to have
the disease, close monitoring of contacts, epidemiological and clinical data collection
from patients, and development of diagnostic and treatment procedures. By Jan 7, 2020,
Chinese scientists had isolated a novel coronavirus (CoV) from patients in Wuhan.
The genetic sequence of the 2019 novel coronavirus (2019-nCoV) enabled the rapid development
of point-of-care real-time RT-PCR diagnostic tests specific for 2019-nCoV (based on
full genome sequence data on the Global Initiative on Sharing All Influenza Data [GISAID]
platform). Cases of 2019-nCoV are no longer limited to Wuhan. Nine exported cases
of 2019-nCoV infection have been reported in Thailand, Japan, Korea, the USA, Vietnam,
and Singapore to date, and further dissemination through air travel is likely.1, 2,
3, 4, 5 As of Jan 23, 2020, confirmed cases were consecutively reported in 32 provinces,
municipalities, and special administrative regions in China, including Hong Kong,
Macau, and Taiwan.
3
These cases detected outside Wuhan, together with the detection of infection in at
least one household cluster—reported by Jasper Fuk-Woo Chan and colleagues
6
in The Lancet—and the recently documented infections in health-care workers caring
for patients with 2019-nCoV indicate human-to-human transmission and thus the risk
of much wider spread of the disease. As of Jan 23, 2020, a total of 835 cases with
laboratory-confirmed 2019-nCoV infection have been detected in China, of whom 25 have
died and 93% remain in hospital (figure
).
3
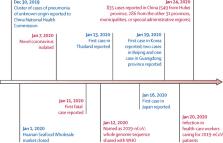
Figure
Timeline of early stages of 2019-nCoV outbreak
2019-nCoV=2019 novel coronavirus.
In The Lancet, Chaolin Huang and colleagues
7
report clinical features of the first 41 patients admitted to the designated hospital
in Wuhan who were confirmed to be infected with 2019-nCoV by Jan 2, 2020. The study
findings provide first-hand data about severity of the emerging 2019-nCoV infection.
Symptoms resulting from 2019-nCoV infection at the prodromal phase, including fever,
dry cough, and malaise, are non-specific. Unlike human coronavirus infections, upper
respiratory symptoms are notably infrequent. Intestinal presentations observed with
SARS also appear to be uncommon, although two of six cases reported by Chan and colleagues
had diarrhoea.
6
Common laboratory findings on admission to hospital include lymphopenia and bilateral
ground-glass opacity or consolidation in chest CT scans. These clinical presentations
confounded early detection of infected cases, especially against a background of ongoing
influenza and circulation of other respiratory viruses. Exposure history to the Huanan
Seafood Wholesale market served as an important clue at the early stage, yet its value
has decreased as more secondary and tertiary cases have appeared.
Of the 41 patients in this cohort, 22 (55%) developed severe dyspnoea and 13 (32%)
required admission to an intensive care unit, and six died.
7
Hence, the case-fatality proportion in this cohort is approximately 14·6%, and the
overall case fatality proportion appears to be closer to 3% (table
). However, both of these estimates should be treated with great caution because not
all patients have concluded their illness (ie, recovered or died) and the true number
of infections and full disease spectrum are unknown. Importantly, in emerging viral
infection outbreaks the case-fatality ratio is often overestimated in the early stages
because case detection is highly biased towards the more severe cases. As further
data on the spectrum of mild or asymptomatic infection becomes available, one case
of which was documented by Chan and colleagues,
6
the case-fatality ratio is likely to decrease. Nevertheless, the 1918 influenza pandemic
is estimated to have had a case-fatality ratio of less than 5%
13
but had an enormous impact due to widespread transmission, so there is no room for
complacency.
Table
Characteristics of patients who have been infected with 2019-nCoV, MERS-CoV, and SARS-CoV7,
8, 10, 11, 12
2019-nCoV
*
MERS-CoV
SARS-CoV
Demographic
Date
December, 2019
June, 2012
November, 2002
Location of first detection
Wuhan, China
Jeddah, Saudi Arabia
Guangdong, China
Age, years (range)
49 (21–76)
56 (14–94)
39·9 (1–91)
Male:female sex ratio
2·7:1
3·3:1
1:1·25
Confirmed cases
835†
2494
8096
Mortality
25† (2·9%)
858 (37%)
744 (10%)
Health-care workers
16‡
9·8%
23·1%
Symptoms
Fever
40 (98%)
98%
99–100%
Dry cough
31 (76%)
47%
29–75%
Dyspnoea
22 (55%)
72%
40–42%
Diarrhoea
1 (3%)
26%
20–25%
Sore throat
0
21%
13–25%
Ventilatory support
9·8%
80%
14–20%
Data are n, age (range), or n (%) unless otherwise stated. 2019-nCoV=2019 novel coronavirus.
MERS-CoV=Middle East respiratory syndrome coronavirus. SARS-CoV=severe acute respiratory
syndrome coronavirus.
*
Demographics and symptoms for 2019-nCoV infection are based on data from the first
41 patients reported by Chaolin Huang and colleagues (admitted before Jan 2, 2020).
8
Case numbers and mortalities are updated up to Jan 21, 2020) as disclosed by the Chinese
Health Commission.
†
Data as of Jan 23, 2020.
‡
Data as of Jan 21, 2020.
9
As an RNA virus, 2019-nCoV still has the inherent feature of a high mutation rate,
although like other coronaviruses the mutation rate might be somewhat lower than other
RNA viruses because of its genome-encoded exonuclease. This aspect provides the possibility
for this newly introduced zoonotic viral pathogen to adapt to become more efficiently
transmitted from person to person and possibly become more virulent.
Two previous coronavirus outbreaks had been reported in the 21st century. The clinical
features of 2019-nCoV, in comparison with SARS-CoV and Middle East respiratory syndrome
(MERS)-CoV, are summarised in the table. The ongoing 2019-nCoV outbreak has undoubtedly
caused the memories of the SARS-CoV outbreak starting 17 years ago to resurface in
many people. In November, 2002, clusters of pneumonia of unknown cause were reported
in Guangdong province, China, now known as the SARS-CoV outbreak. The number of cases
of SARS increased substantially in the next year in China and later spread globally,
14
infecting at least 8096 people and causing 774 deaths.
12
The international spread of SARS-CoV in 2003 was attributed to its strong transmission
ability under specific circumstances and the insufficient preparedness and implementation
of infection control practices. Chinese public health and scientific capabilities
have been greatly transformed since 2003. An efficient system is ready for monitoring
and responding to infectious disease outbreaks and the 2019-nCoV pneumonia has been
quickly added to the Notifiable Communicable Disease List and given the highest priority
by Chinese health authorities.
The increasing number of cases and widening geographical spread of the disease raise
grave concerns about the future trajectory of the outbreak, especially with the Chinese
Lunar New Year quickly approaching. Under normal circumstances, an estimated 3 billion
trips would be made in the Spring Festival travel rush this year, with 15 million
trips happening in Wuhan. The virus might further spread to other places during this
festival period and cause epidemics, especially if it has acquired the ability to
efficiently transmit from person to person.
Consequently, the 2019-nCoV outbreak has led to implementation of extraordinary public
health measures to reduce further spread of the virus within China and elsewhere.
Although WHO has not recommended any international travelling restrictions so far,
15
the local government in Wuhan announced on Jan 23, 2020, the suspension of public
transportation, with closure of airports, railway stations, and highways in the city,
to prevent further disease transmission.
16
Further efforts in travel restriction might follow. Active surveillance for new cases
and close monitoring of their contacts are being implemented. To improve detection
efficiency, front-line clinics, apart from local centres for disease control and prevention,
should be armed with validated point-of-care diagnostic kits.
Rapid information disclosure is a top priority for disease control and prevention.
A daily press release system has been established in China to ensure effective and
efficient disclosure of epidemic information. Education campaigns should be launched
to promote precautions for travellers, including frequent hand-washing, cough etiquette,
and use of personal protection equipment (eg, masks) when visiting public places.
Also, the general public should be motivated to report fever and other risk factors
for coronavirus infection, including travel history to affected area and close contacts
with confirmed or suspected cases.
Considering that substantial numbers of patients with SARS and MERS were infected
in health-care settings, precautions need to be taken to prevent nosocomial spread
of the virus. Unfortunately, 16 health-care workers, some of whom were working in
the same ward, have been confirmed to be infected with 2019-nCoV to date, although
the routes of transmission and the possible role of so-called super-spreaders remain
to be clarified.
9
Epidemiological studies need to be done to assess risk factors for infection in health-care
personnel and quantify potential subclinical or asymptomatic infections. Notably,
the transmission of SARS-CoV was eventually halted by public health measures including
elimination of nosocomial infections. We need to be wary of the current outbreak turning
into a sustained epidemic or even a pandemic.
The availability of the virus' genetic sequence and initial data on the epidemiology
and clinical consequences of the 2019-nCoV infections are only the first steps to
understanding the threat posed by this pathogen. Many important questions remain unanswered,
including its origin, extent, and duration of transmission in humans, ability to infect
other animal hosts, and the spectrum and pathogenesis of human infections. Characterising
viral isolates from successive generations of human infections will be key to updating
diagnostics and assessing viral evolution. Beyond supportive care,
17
no specific coronavirus antivirals or vaccines of proven efficacy in humans exist,
although clinical trials of both are ongoing for MERS-CoV and one controlled trial
of ritonavir-boosted lopinavir monotherapy has been launched for 2019-nCoV (ChiCTR2000029308).
Future animal model and clinical studies should focus on assessing the effectiveness
and safety of promising antiviral drugs, monoclonal and polyclonal neutralising antibody
products, and therapeutics directed against immunopathologic host responses.
We have to be aware of the challenge and concerns brought by 2019-nCoV to our community.
Every effort should be given to understand and control the disease, and the time to
act is now.
This online publication has been corrected. The corrected version first appeared at
thelancet.com on January 29, 2020
Related collections
Most cited references2
- Record: found
- Abstract: found
- Article: not found
The epidemiology of severe acute respiratory syndrome in the 2003 Hong Kong epidemic: an analysis of all 1755 patients.
Gabriel Leung, Anthony J. Hedley, Lai-Ming Ho … (2004)
- Record: found
- Abstract: found
- Article: not found
Age- and Sex-Specific Mortality Associated With the 1918–1919 Influenza Pandemic in Kentucky
Cecile Viboud, Jana Eisenstein, Ann Reid … (2012)
