- Record: found
- Abstract: found
- Article: found
Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices—United States, 2018–19 Influenza Season
Read this article at
Summary
This report updates the 2017–18 recommendations of the Advisory Committee on Immunization Practices (ACIP) regarding the use of seasonal influenza vaccines in the United States (MMWR Recomm Rep 2017;66[No. RR-2]). Routine annual influenza vaccination is recommended for all persons aged ≥6 months who do not have contraindications. A licensed, recommended, and age-appropriate vaccine should be used. Inactivated influenza vaccines (IIVs), recombinant influenza vaccine (RIV), and live attenuated influenza vaccine (LAIV) are expected to be available for the 2018–19 season. Standard-dose, unadjuvanted, inactivated influenza vaccines will be available in quadrivalent (IIV4) and trivalent (IIV3) formulations. Recombinant influenza vaccine (RIV4) and live attenuated influenza vaccine (LAIV4) will be available in quadrivalent formulations. High-dose inactivated influenza vaccine (HD-IIV3) and adjuvanted inactivated influenza vaccine (aIIV3) will be available in trivalent formulations.
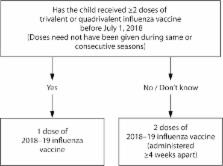
Updates to the recommendations described in this report reflect discussions during public meetings of ACIP held on October 25, 2017; February 21, 2018; and June 20, 2018. New and updated information in this report includes the following four items. First, vaccine viruses included in the 2018–19 U.S. trivalent influenza vaccines will be an A/Michigan/45/2015 (H1N1)pdm09–like virus, an A/Singapore/INFIMH-16-0019/2016 (H3N2)-like virus, and a B/Colorado/06/2017–like virus (Victoria lineage). Quadrivalent influenza vaccines will contain these three viruses and an additional influenza B vaccine virus, a B/Phuket/3073/2013–like virus (Yamagata lineage). Second, recommendations for the use of LAIV4 (FluMist Quadrivalent) have been updated. Following two seasons (2016–17 and 2017–18) during which ACIP recommended that LAIV4 not be used, for the 2018–19 season, vaccination providers may choose to administer any licensed, age-appropriate influenza vaccine (IIV, RIV4, or LAIV4). LAIV4 is an option for those for whom it is appropriate. Third, persons with a history of egg allergy of any severity may receive any licensed, recommended, and age-appropriate influenza vaccine (IIV, RIV4, or LAIV4). Additional recommendations concerning vaccination of egg-allergic persons are discussed. Finally, information on recent licensures and labeling changes is discussed, including expansion of the age indication for Afluria Quadrivalent (IIV4) from ≥18 years to ≥5 years and expansion of the age indication for Fluarix Quadrivalent (IIV4), previously licensed for ≥3 years, to ≥6 months.
This report focuses on the recommendations for use of vaccines for the prevention and control of influenza during the 2018–19 season in the United States. A Background Document containing further information and a brief summary of these recommendations are available at https://www.cdc.gov/vaccines/hcp/acip-recs/vacc-specific/flu.html.
These recommendations apply to U.S.-licensed influenza vaccines used within Food and Drug Administration–licensed indications. Updates and other information are available at CDC’s influenza website ( https://www.cdc.gov/flu). Vaccination and health care providers should check CDC’s influenza website periodically for additional information.
Related collections
Most cited references64
- Record: found
- Abstract: found
- Article: not found
Efficacy of high-dose versus standard-dose influenza vaccine in older adults.
- Record: found
- Abstract: found
- Article: not found
Randomized, double-blind controlled phase 3 trial comparing the immunogenicity of high-dose and standard-dose influenza vaccine in adults 65 years of age and older.
- Record: found
- Abstract: found
- Article: not found